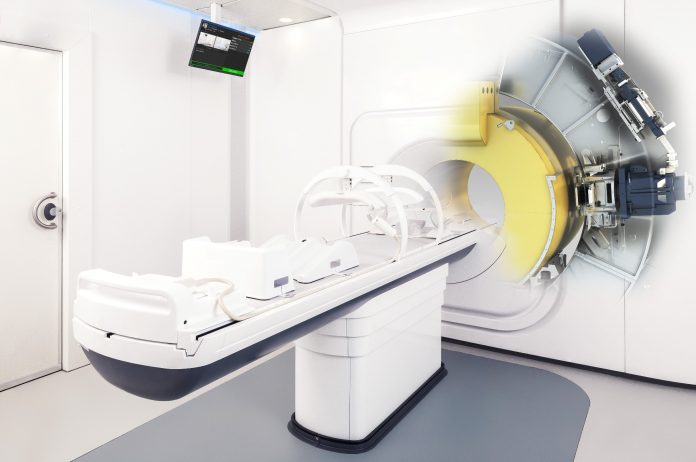
A revolutionary new form of radiotherapy at The Christie has been given the green light from European regulators to begin treating patients.
Elekta Unity, the world’s first high-field MR-linac, received it’s CE mark approval last week enabling commercial sales and clinical use in Europe.
The Christie is one of just seven founding research partners in the MR-linac consortium, helping to ensure that Christie patients are some of the first in the world to benefit from this pinpoint pioneering new radiotherapy technology.
The MR-guided linear accelerator (MR-linac) combines magnetic resonance (MR) scanning and tumour-busting radiotherapy to deliver magnetic resonance radiotherapy in one hi-tech package.
The installation of the machine at The Christie was completed last year, with the first item (a red rose) being scanned in October. The awarding of the CE mark is one of the final milestones in the project before it is used to treat patients.
Magnetic resonance radiotherapy is an advanced form of radiotherapy that will potentially enable The Christie to deliver more targeted treatment for patients with certain cancers.
MR-linac can precisely locate tumours, tailor the shape of X-ray beams in real time, and lock-on to tumours during treatment, to deliver radiotherapy even when tumours are moving.
It can also see if a tumour changes shape, location or size between treatment sessions.
Planning radiotherapy in “real time” avoids the need to rely on MRI scans that have been taken at an earlier date.
The next major milestone for the MR-linac project will be the first scans of a human body which will be undertaken on a volunteer.
Dr Ananya Choudhury, clinical lead for MR-linac at The Christie, said: “This is a very exciting milestone in this flagship research project for The Christie. We expect the MR-linac to deliver more precise, more personalised, more effective and kinder radiotherapy to our patients. It lets us see tumours very clearly and treat them at the same time with pinpoint accuracy.”
Richard Hausmann, President and CEO of Elekta added: “Receiving CE mark for Unity is a big achievement in revolutionising the field of radiation therapy and a real watershed moment for everyone involved in treating cancer patients. The change that MR/RT will bring to cancer therapy is paramount in advancing patient care and outcomes. The Christie has played a critical role in bringing this transformative therapy to the clinic, and I’m thankful for their contributions as well as those of the other MR-linac consortium members, Philips (our MR technology partner) and our dedicated employees. Their dedication and commitment to meeting the needs of patients and clinicians helped us to reach this important day.”
Fundraisers at The Christie charity have contributed £1m towards the overall cost of the MR-linac.
It is hoped that the first patient treatment, as part of a trial, will be available at The Christie early 2019.
The Christie is working with other leading international cancer centres in North America and Europe as part of the Elekta MR-linac consortium. The MRI technology partner for Elekta is the global electronics giant Philips.
Bringing MR-linac to The Christie, along with proton beam therapy, will ensure that the hospital remains at the forefront of leading-edge radiotherapy treatment and research. The Christie will be the only NHS hospital in the UK to offer both MR-linac and proton beam therapy by the end of this year.